Mastering Restaurant Sanitizing: Best Practices for Kitchens and Dining Areas
Master restaurant sanitizing with clear steps for kitchens and dining areas. Learn methods, checklists, and procedures to keep your team safe and...
FSMA regulations aim to shift the focus of manufacturers and governing agencies toward preventing foodborne illnesses.
During President Barack Obama's first term, he signed the FSMA into law, giving the U.S. Food and Drug Administration (FDA) new authority to regulate the growing, harvesting, and processing of food.
FSMA stands for the Food Safety Modernization Act.
In short, the United States' FSMA is a law that regulates the production and distribution of food with an increasing focus on traceability. The Food Safety Modernization Act is composed of several sections that address the different concerns of each sector in the food industry.
Ultimately, the FSMA strives to minimize the potential impacts of foodborne illnesses and protect the economic well-being of the nation's food system. Since the regulation's signing into law, it has been continuously developed to encompass a broader range of the industry and includes the whole supply chain.
The Food Safety Modernization Act was passed in response to the surge in significant foodborne illness outbreaks and heightened bioterrorism concerns following the events of 9/11 in 2001.
This event prompted the United States government to create a law that granted the FDA the capability to impose detention over food for consumption that has questionable integrity and may pose a threat to people or animals.
In 2009, Congress and the Obama Administration introduced new food safety measures which extended regulations to include farm-level operations.
If your a food handler and you see this on a food safety test, the FSMA was passed because:
Former U.S. President Obama signed the FSMA into law on January 4, 2011. Although the law and its many rules were not finalized until after several years of signing, some consider it "the most sweeping reform of our food safety laws in more than 70 years."
The purpose of the FSMA is primarily to strengthen the United States food safety system and supply. It does this by focusing on three fundamental strategies: prevention, increased surveillance, and better response and recovery from raw material production and supply to the distribution of products.
What these areas of focus allow food businesses (and the agencies which govern them) to do is act — when necessary — proactively instead of reactively, which is in the best interest of public health.

In this section, we discuss the 7 rules of FSMA:
To make the law more efficient and effective, seven major rules were designed to ensure clear resolutions to address food safety concerns. Below are brief explanations and an overview of these rules.

This safety-produce rule states that science-based minimum standards for food safety will be set for food safety practices, including farm manufacturing, growing, harvesting, post-harvest controls, and holding of produce for human consumption. Products include fruits and vegetables grown for human consumption at the farm level.
The FDA rule requires farms to perform an initial survey on their own farm practices and material sources.
All practices intended to result in the production of food for humans are regulated under this provision. This rule also requires increasing worker education in health and hygiene by undergoing food safety training.
While the agency provides educational programs on FSMA rules, the responsibility of training employees on food safety is a major job for business owners.
The Foreign Supplier Verification Program rule regulates the possibilities of hazards from adulteration and misbranding, such as the undeclared presence of allergens. Some of the specific scopes and enforcement actions of this FSMA regulation include:
This rule under the Food Safety Modernization Act aims to prevent food safety issues concerning improper sanitary conditions during transport. It emphasizes the protection of food during transportation against contamination or spoiling while in transit. Some provisions include applicable requirements for food refrigeration, sanitation of vehicle and food contact surfaces in between loads, and proper recording of these conditions. Note that this rule does not apply to food supply being shipped through the air due to limitations of the law.
This rule applies to logistics operations, including carriers by motor or rail vehicles as well as the receivers of food products.
Under the sanitary transportation rule, the following standard regulatory requirements are needed to be established by food handlers:
In addition, compliance with basic food safety practices, such as the current Good Manufacturing Practices (cGMP) for covered facilities, would significantly help in complying with the Food Safety Modernization Act. Stricter implementation of training for employee education on food safety is also part of the policy.
All company employees are mandated to be oriented and familiarized with FSMA preventive controls requirements. This includes allergen management requirements and cross-contact issues to prevent the risk of food allergy. The law mandates businesses to agree to the significant authority of the FDA to suspend operations once transportation of food under conditions that are unsafe for humans is reported.
The FSMA has considered acts of terrorism concerning adulterated food. Unfavorable personnel actions can cause serious health issues and economic disturbances.
As such, the FSMA requires concerned businesses to build and implement a Food Defense Plan containing mitigation strategies on how to prevent acts of terrorism and evaluate vulnerable points of the food processing line.
This strategic document aims to address the probability of an outbreak due to potential food safety risks of intentional adulteration and any form of food supply chain-targeted terrorism.
This FSMA rule focuses on prevention strategies by gaining more information on potentially high-risk processes in a food production line. It applies to foreign and local food manufacturers producing large volumes of products that are required to report to the FDA; thereby, small businesses, including farms, are not bound by it.
The main components and procedures for Food Defense include:
The mandated time of compliance with this rule depends on the size of the firm. That is, very small businesses, small businesses, and others are categorically required to comply five, four, and three years after the publication of the rule, respectively.
This rule aims to establish volunteer third-party certification bodies to monitor and evaluate foreign firms. Once certified, auditors and subject matter experts are given the authority to perform consultations in preparation for regulatory audits.
The certification acquired from this rule can be used to participate in the FDA's Voluntary Qualified Imported Program (VQIP) to gain expedited reviews for food entry.
The regulation covers legal authorities, competency, conflict-of-interest conditions, and quality assurance steps for entities seeking to become accredited auditors and for companies who want to be accredited. Accreditation bodies such as your company will be given the authority to perform the following tasks and report to the FDA:
This rule, as stated by the FDA, contains competency, capacity, conflict-of-interest safeguards, and legal authority as applicable requirements for accreditation.
Accredited certification bodies will be allowed to perform both consultative and regulatory accreditations. The former type is an auditing activity to prepare a company for regulatory food safety audits and internal use. It evaluates the suitability of a company being accredited based on industry standards. Accredited certification bodies can be foreign governments.
The FSMA regulation on preventive control requirements applies to both foods for human and animal consumption. This federal rule requires producers to have a comprehensive hazard analysis program and establish comprehensive, science-based preventive controls for these standards.
This rule under the FSMA regulations contains amendments to the previous provisions of the current good manufacturing practices. One of the most notable amendments is the legislative mandate to conduct education and training for food-handling employees, which was previously not binding. Training requirements involve the basic principles of food hygiene and food safety, as well as the importance of their health status and food handling practices.
In terms of animal health and food safety control, FSMA regulations and enforcement policy require food handlers processing animal by-products to make as feeds must adhere to current Good Manufacturing Practices (cGMP). Farmers can choose whether to use their preexisting cGMP regulations for human foods or create an entirely new plan for animal foods.
The preventive control rule requires food processing owners to have a comprehensive food safety plan. This document will be a food safety management system composed of the following components:
The VQIP is a fee-based program provided by the FDA FSMA to foreign food facilities that intend to import their products to the country. The main purpose of this FSMA rule is to speed up the process of inspection and evaluation of their products, whether intended for human or for animal consumption.
Requirements for safe food certifications under the VQIP ensure that the importer adheres to the food safety regulations of the country. This also assures the consumers that the imported products have passed the local food safety regulatory requirements.
The critical provisions of FSMA requirements emphasize the following:
FSMA compliance means having a strong and working Food Safety Management System (FSMS) in your company. We will help you understand and implement top-quality FSMS guidelines and give you an overview of FSMA compliance.

Under the FDA Food Safety Modernization Act, the strictness of monitoring and imposing foreign and domestic supplier verification programs have been significantly strengthened to protect the public. The FDA is given approximately 50 regulations and guidelines for businesses operating and those with transactions in the country.
With exemptions to some industries, FSMA compliance requires all food processors to create a risk-based hazard analysis program. Within the food processing flow, a manufacturing company must create a preventive control program for significant and foreseeable hazards under the FSMA.
The FSMA also promotes the use of scientific methods to establish regulations on food safety. It specifically requires all concerned industries to comply and create an FSMA food safety plan, hazard and critical control point identification, the establishment of controls and preventive food safety measures, monitoring, recall plan, and more stringent documentation of inspection reports.
This law emphasizes the authority of the FDA to impose recalls on contaminated foods or products that do not adhere to the rules of the law. It has also mandated the FDA to require involved businesses to submit to new preventive protocols and safety plans. Additionally, the Food Safety Modernization Act increased the capacity of the FDA to inspect businesses and their authority to oversee overseas transactions.
Under the newly reformed rules of the FDA and as a mandate from the Food Safety Modernization Act, food manufacturers are required to submit and register with the FDA every two years as compliance, according to Section 415 of the Federal Food, Drug, and Cosmetic Act (FD&C Act) as amended by FSMA.
In addition, the FDA requires businesses to create a Food Safety Plan as well as a Food Defense Plan that is science-based and come up with preventive control strategies for hazards and potential vulnerabilities.
The Food Safety Modernization Act also includes a rule for the FDA to establish laboratory testing regulations to promptly detect and respond to food safety issues.
The FSMA rules require all departments of health and human services to create regulations on the safe harvesting and handling of raw materials, importation of foods, and increased inspections. To promote the preventive approach, manufacturers and suppliers are required by this law to provide a detailed list of corrective actions to the identified hazards as well as a Food Defense plan.
Compliance with the Food Safety Modernization Act does not only mean registering with the FDA. It also means complying with the rules they set out and submitting essential plans.
As an example, making a food safety plan would require you to submit a report of your whole processing functions and create a plan with the following elements:
All food manufacturers and suppliers who are regulated by the FDA must comply with FSMA regulations in order to help protect consumers from potential food safety hazards. Included in this group are all domestic and foreign food manufacturers and suppliers whose products are in the United States as well as food and beverage companies with an implemented HACCP plan.
This accounts for about 78% of the U.S. food supply, leaving the other 22% under the care of the USDA.
At a high-level, the groups that fall under FSMA regulations and achieve (and maintain) compliance include:

This means that all domestic facilities and foreign food establishments that have transactions with the country must comply with the FSMA. Food industries dealing with fresh produce, such as meat, dairy products, and poultry, that are under the USDA are not included in the provisions of the FSMA.
If you are a commercial farm owner in the food packaging label industry or food processing industry, the FSMA applies to your business.
Under the completed FSMA, different time frames and compliance dates have been provided to allow concerned businesses to comply. Because FSMA is a law, all businesses under the regulations of the FDA are mandated to adhere to its provisions.
Non-compliance to the provided rules may put the safety and intent of consumers at risk. Because the FSMA aims to reduce hazards and risks in a food company's production, failure to comply can result in overlooking potential contaminations and breaches of food safety protocols.
The following are some of the potential risks of non-compliance to your business and consumers:
In cases of detection of adulteration in articles of food, food recall activities are rightfully issued by the FDA. Non-compliance with the FSMA rule regarding intended adulteration can lead to the suspension of your company's registration. This removes the company's authorization to legally produce and sell its products for a period of time.
For less severe issues with non-compliance to the FSMA, re-inspection of physical locations and reevaluation of guidance documents may be imposed by the FDA. This kind of process entails fees that can be charged to your company and are currently at around $231 to $285 per hour.
Other examples of violation and non-compliance to FSMA include refusal to comply with recalling orders, inadequate FSMA Food Safety Plan and Food Defense Regulations, and importing products from suppliers without a risk-based evaluation of their food product safety. To an extent, criminal penalties for non-compliance can include fines of up to $250,000 should serious injuries incur from the issue.
This kind of case can also lead to temporary restrictions on operations that can lead to a significant loss in revenues. In extreme cases, those which involve the death of a consumer or serious hospitalization due to non-compliance, companies can be shut down for good.
In this section, we discuss some of the other regulations included in the FSMA:
In addition to the 7 major FDA FSMA regulations, the regulatory agency has also amended some of the preexisting regulations to strengthen its food safety regulation aspect.
Included in these related programs are proposed new federal food safety laws that are undergoing discussions and reviews by the related government agencies and health departments. Currently, some of the proposed food regulations in this list are yet to be approved to take effect.
The temporary policy on food traceability was approved in November 2022. This FSMA rule serves as an additional requirement to the previously established traceability program of the FDA.
This amendment is considered one of the key requirements of the FDA's New Era of Smarter Food Safety Blueprint. It aims to devise a way to easily track down recipients of contaminated products and effectively mitigate the chances of an outbreak.
The center of this amendment is the additional recordkeeping requirement for Key Data Elements associated with different Critical Tracking events analyzed by the FDA.
According to the FSMA 204, manufacturers who process and handle foods under the Food Traceability List (FTL) are required to comply with this rule. The FTL is a peer-reviewed list of high-risk foods and was generated by a risk-ranking model developed by the FDA.
The list of foods was generated from a model which counted the frequency of outbreaks, the severity of potential illnesses, the likelihood of contamination, and other hazard-based criteria.
Among the FSMA regulations, this rule aims to establish a program for the microbial and chemical testing of foods by laboratories. The rule will require food analysis to be conducted by laboratories that have voluntarily applied for evaluation by accreditation bodies.
Laboratories accredited under this program will be given the authority to give evidence in cases of questionable food items, analyze food items under suspicion of a food safety hazard, respond to food testing orders by the FDA, and address any identified import alert related to food safety.
This final FDA FSMA rule is an amendment to a preexisting rule on the food safety requirements for food processing facilities registration. It aims to collect more information and build a more accurate registration base for food companies reporting under the FDA.
Some of the notable revisions include requiring an email address for registration, a mandatory renewal of your processing facility registration every two years, and giving the FDA absolute permission to conduct an inspection as stated by the guidelines of the Federal Food, Drug, and Cosmetic Act.
The purpose of this final FSMA rule is to expand the access of the FDA to records related to foods that have shown a reasonable possibility of food safety risks.
In addition, the FDA is also granted access to the records of other food products from a single company that they believe has the possibility of having the same potential risk.
Under this final rule, importers of human and animal food are required to submit additional requirements, which will serve as prior notice. The notice will include information if the product has been previously refused entry by a different country and requires the importer to declare this country.
The rule strengthens the FDA's resolve to address any potential food safety risks even before they happen. In this case, even before they enter the country.
The approval of this final rule has given the FDA stronger authority to recall, detain, and analyze any food articlethat has shown even just a slight potential of having food safety hazards. Reasons for recall may be signs of adulteration or misbranding.
More detailed information on the FSMA Rules, including final compliance dates and enforcement action, is available on the FDA central source website.
Inspection by the FDA can come at any time. This fact means that your business must have a solid and working food safety system at all times. Your food safety plan must have a proactive approach to tackle potential food hazards.
For your food business to ensure FSMA compliance, a set of documented processes needs to be prepared. An established food safety plan contains elements consisting of identified food safety hazards, activities that ensure food safety, corrective and recall plans, and others.
Below, we give you a checklist of what you need to comply with the FSMA:
FSMA compliance requires your business to create a product and facility plan, which includes hazard identification, established critical control points, and specifications. This plan includes information about your product, such as its intended use and ingredients.
Hazards are properly identified as a result of raw materials, manufacturing practices, sanitation controls for food contact surfaces, and equipment maintenance procedures. Your food safety plan must also include a proper risk assessment to determine the severity of the identified hazards.
Identified and analyzed hazards must also be accompanied by a set of preventive controls to control them. FSMA compliance requires more than the usual preventive controls for critical control points. Under the FSMA ruling, preventive controls for processes, supply chains, allergens, sanitation procedures, and recall procedures are also required.
Uunder the FSMA, your company is also required to ensure that the suppliers you are dealing with apply equally strict food safety plans. Your suppliers are required to submit proof that they conduct a proper hazard analysis and apply adequate preventive controls. They must secure supply chain program requirements and co-manufacturer supplier approval documents.
To maintain the high integrity of your food safety plan, a well-structured recall strategy is required. This plan will contribute to FSMA compliance by presenting steps on how to address and perform recalls by the responsible party.
Steps may include notification of consignees of the article of food in question, potential directives on how to dispose or return contaminated food, notifying the public about the potential leak of contaminated products, and verifying the effectiveness of the plan.
The plan must be set to immediately control the further release of the food products in question and quickly retrieve batches that are already circulating the market.
This program will allow food business owners to keep track of their material and product's movement throughout the supply chain. It is based on the additional recordkeeping requirements of the new FSMA FDA rule.
The system must be able to capture information about the product, such as the formulation or recipe used, production batch number, the quantity of the batch, destination, and maximum shelf-life.
To provide proof that the current FSMA food safety plan implemented in your business is effective, you need records of monitoring and verification. Reports, records, and forms on tracking the progress and application of the food safety plan must always be ready in case of an audit of company practices.
This checklist only proves that FSMA compliance requires your business and all personnel to have all of their hands on deck and participate in making a well-rounded food safety plan.


Thank you for downloading!
Want to get a customizable HACCP template?
Or set up your food safety system in 15 minutes?
An FSMA compliance statement outlines what regulations you've covered in your facility. It includes the FSMA you're compliant with and documents such as your HACCP plan, recall plan, allergen management plan, and more.
Once your food business has ticked most of the boxes in your FSMA compliance checklist, your business is required to create an FSMA compliance statement. This document states which conditions and rules of the FSMA have your company already complied with.
The FSMA compliance statement is used to list which of the seven major rules and related programs your business is compliant with. Along with the rules are the compliance dates when your food business has fully complied. It must also contain which among the food safety plan checklist you have fulfilled (e.g., hazard analysis, preventive controls, recall plan, etc.).
This in-house document does not have to be too technical, but it must be forwarded if asked as proof of your FSMA compliance. Your FSMA compliance statement is somehow similar to what the Preparedness and Response Act of 2002 (Bioterrorism Law) requires you to provide your customers. Upon request or delivery of some products, this document must be provided.
In line with the FDA's objective to transform the nation's food safety system, the agency initiated extensive partnerships with related government and private organizations. The partnerships mainly aimed to create training programs for food business owners and food producers to help them comply with the FSMA requirements.
While it is every business owner's responsibility to seek the necessary training for FSMA, the FDA and its partner organizations launched training opportunities for farm operators, food processors, retail merchants, and wholesalers.
The Food Safety Preventive Controls Alliance (FSPCA), a partner organization of the FDA, developed an educational training program for domestic and foreign food industries. The program includes distance training modules with curricula that mainly involves the following:
The FDA is also working with corporate agreements to develop training programs fit for the other businesses in the industry. The programs aim to promote industry understanding of the law. Since 2015, the FDA has collaborated with the USDA National Institute of Food and Agriculture to provide grant programs to eligible food business owners.
To date, the FDA and its partner organizations are providing training programs, paid and free, to help familiarize and prepare business owners with the regulatory requirements of the food law.
The FSMA applies to businesses that are categorized as commercial farms, including farm packing operations and food processing facilities. Under each FSMA final rule, specific guidelines for training requirements are outlined.
At least one trained individual must be present in a food facility. In particular, every food facility is required to have a preventive controls qualified individual (PCQI). A PCQI is a trained regulatory personnel who has received training on performing risk assessments and developing a food safety plan.
Food businesses must also note that the FDA specifically reviews the suitability of the facility's food safety plan and not the qualifications of an assigned PCQI.
Complying with the FSMA and food safety systems regulations means having a strong Food Safety Management System in your business's central location for distribution and branches as one of the many requirements.
At FoodDocs, our objective is for every food business to become food safety compliant. Our digital Food Safety Management System was built by food safety experts who have seen the food industry evolve even further. Using our system, you can get a comprehensive food safety management software in 15 minutes.
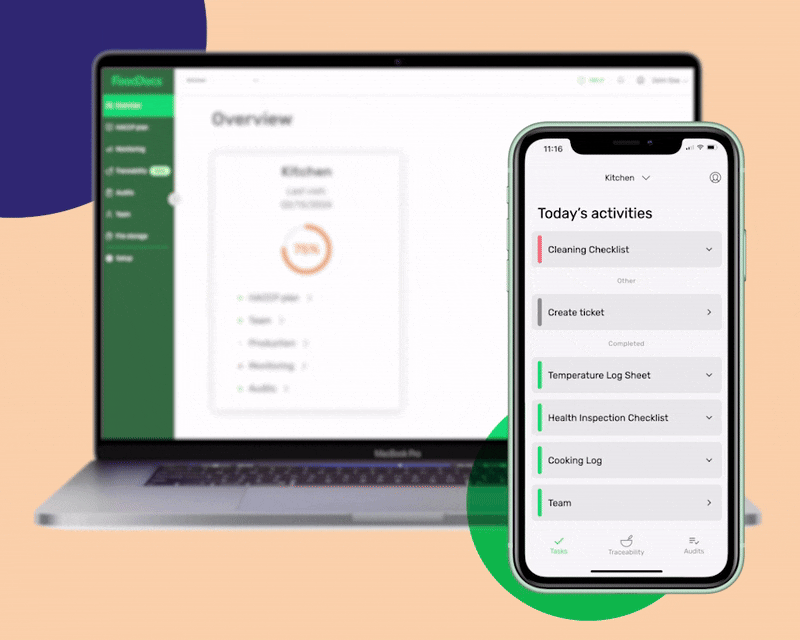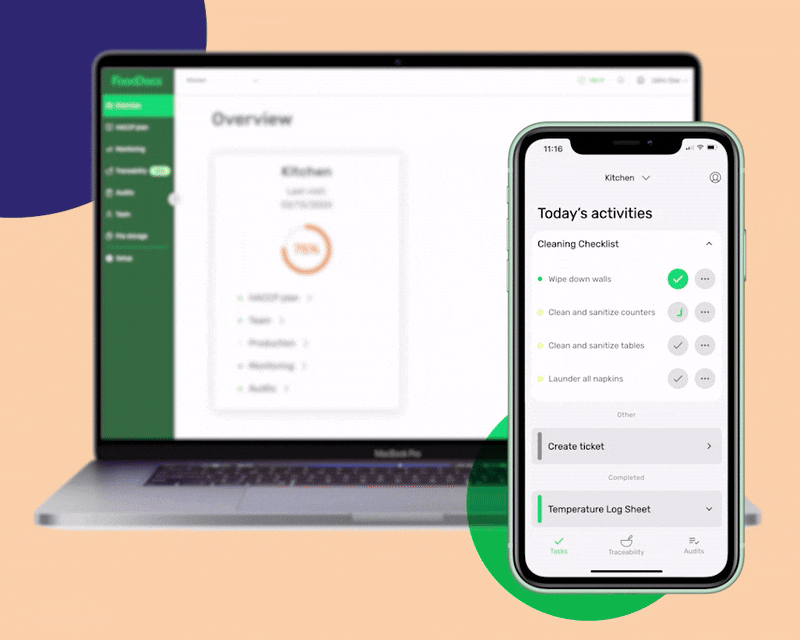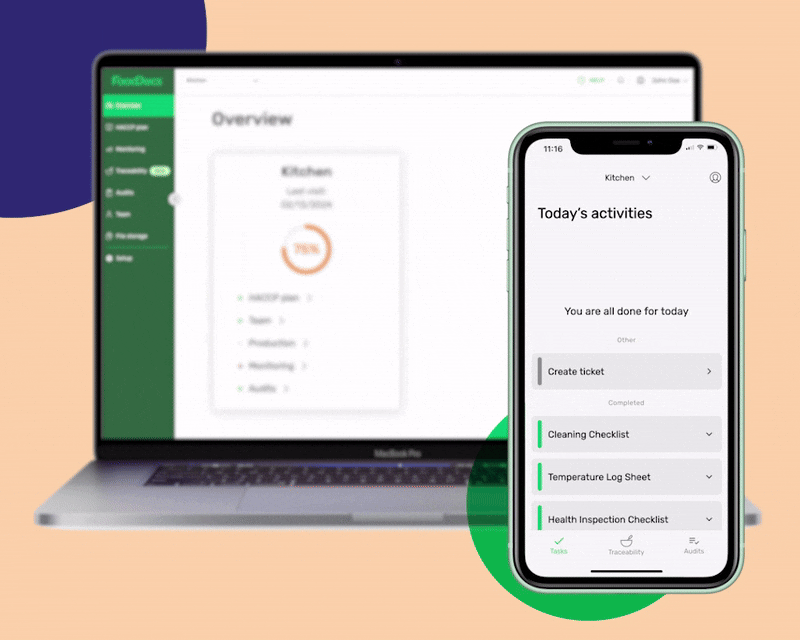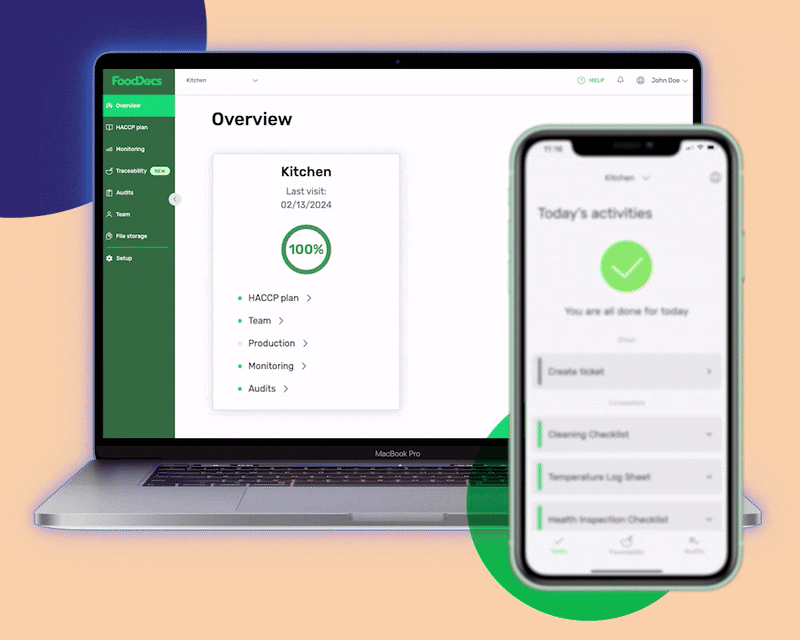
An all-in-one digital food safety management system, complete with a HACCP plan builder, Monitoring system, and Food Traceability software.
Starting with Traceability, our system is:
Setting up an internal traceability system is a continuous task and requires serious commitment from your food safety team. Using FoodDocs' smart Food Safety Management System equipped with a two-in-one Traceability System fulfills all traceability and production needs.

Moving on to Monitoring, you'll get:
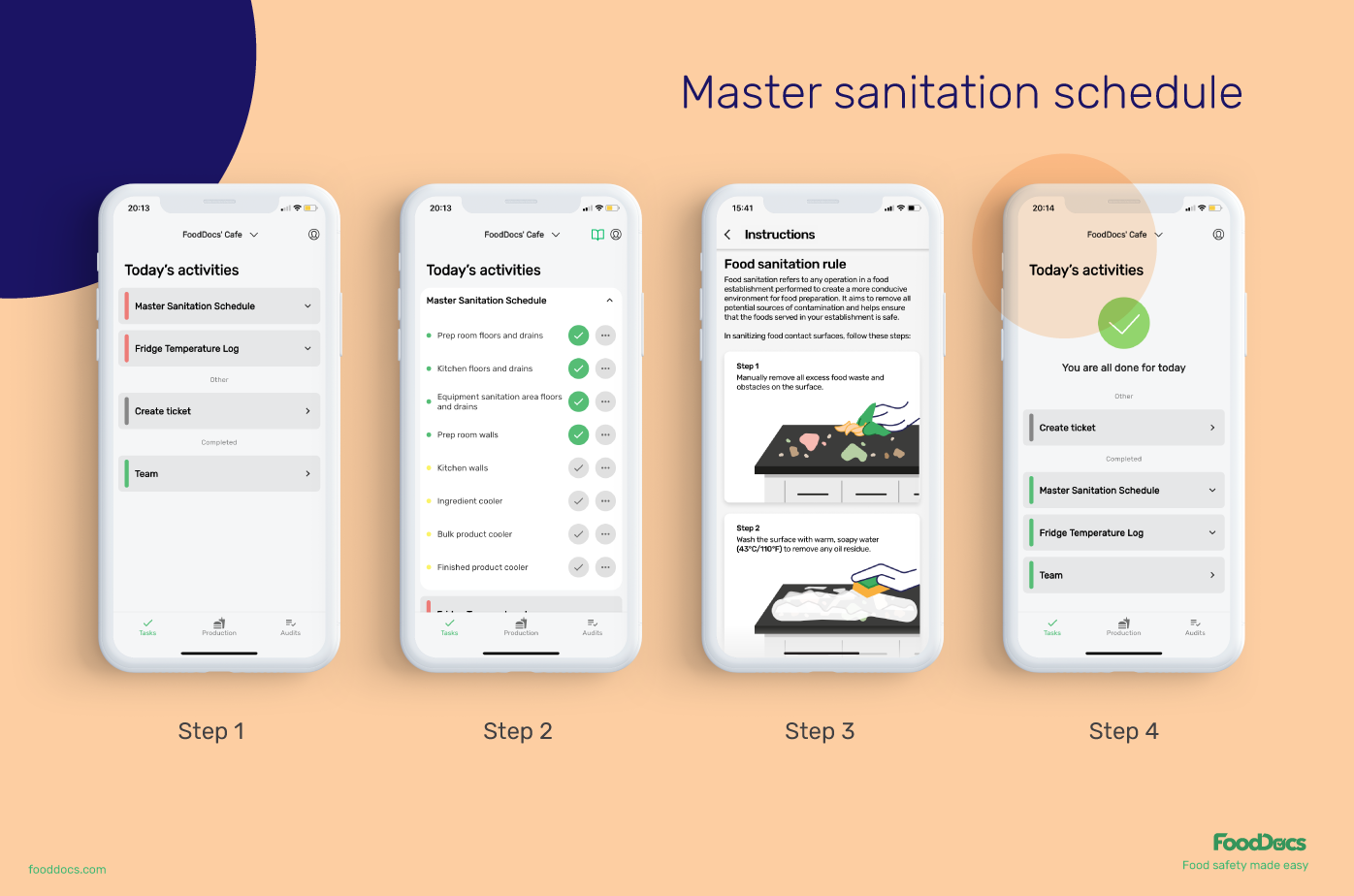
In addition to features that will help food employees fulfill every food safety task, our digital FSMS can also help managers improve the efficiency of their operations.
The FSMA food safety plan is a comprehensive document that consists of preventive controls and a systematic approach for identifying and analyzing food safety hazards. The plan is established to minimize the likelihood of causing foodborne illnesses as a result of consuming your products.
All food producers, growers, processors, and distributors of high-risk imported foods and domestic products regulated by the FDA are required to comply with the key requirements of FSMA for public health protection.
Generally, HACCP and FSMA are not the same. The HACCP is a food safety system that is used to identify, evaluate, and control food safety hazards, whereas the FSMA is a law that contains preventive controls (HARPC) for the production, processing, transportation, and import of foods in the US food chain.
The FDA is the main implementing regulatory agency of the FSMA law. This means that all food establishments that are regulated by the FDA must comply with the FDA FSMA regulations, which account for at least 75% of the food industry.
The seven major rules of the FDA FSMA include the following:
All responsible parties and food businesses registered under the FDA are required to have a food safety plan that is based on the requirements of the preventive controls rule for human food.
The FSMA gave the U.S. Food and Drug Administration more power to control how millions of food products are produced, collected, and processed, as well as enforce sanctions in the supply chain to achieve the national food safety goals of public health protection. This law also provides protections and improvements in food safety operations in the food industry, including the authority to detain suspect food in foodborne illness cases.
Despite not applying to any restaurant and retail food establishment, some of the FSMA rules, such as the Sanitary Transportation Rule, may apply. The preventive food safety measures and food requirements of the FSMA can prove to be more than useful and effective in achieving human health goals for the restaurant industry.
The major highlights of the regulatory requirements of the FDA Food Safety Modernization Act include five key elements:
The FDA Food Safety Modernization Act does not have authority over food items that include meat, poultry, dairy, and related products produced by human and animal food facilities. Qualified facilities handling this food group are regulated by the enforcement discretion of the USDA.
Not all rules and sub-provisions of the FSMA include food packaging industries. In general, any manufacturing practice related to food packaging is not regulated by the FDA FSMA rules.
The FSMA strengthens the authority of the FDA and expands its capabilities for public health protection from the malpractice of food producers. The law focuses on preventing food safety issues from happening as a result of providing imported and domestic products rather than reacting to them to save more healthy recipients of food from any foodborne illness.
The Accredited Third-Party Certification Rule provides a voluntary program for evaluating and recognizing entities that will conduct third-party certification for foreign facilities. The FSMA certification program aims to ensure the competency of the accredited third-party auditors under the program.

Master restaurant sanitizing with clear steps for kitchens and dining areas. Learn methods, checklists, and procedures to keep your team safe and...

Learn challenges healthcare foodservice teams face today and key food safety practices to protect vulnerable patients. Get a free healthcare leader...

Learn what Standard Operating Procedures (SOPs) are and how to write effective SOPs that ensure consistency, efficiency, and safety in your...